Tirzepatide
Glucagon-like Peptide-1 (GLP-1) Receptor Agonist and Glucose-dependent Insulinotropic Polypeptide (GIP)
- Mounjaro™
- LY3298176
Contents
Most Frequent Uses:
- Tirzepatide is a glucagon-like peptide 1 (GLP-1) receptor agonist AND a glucose-dependent insulinotropic polypeptide (GIP) indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus
- As monotherapy when metformin is inappropriate due to contraindication or intolerance
- Weight loss support along with diet and exercise
- Nonalcoholic steatohepatitis
Dosage:
- 15mg/ml and 18mg/ml
For T2D or pre-diabetes – SubQ
- Start at 2.5 mg SQ once weekly.
- After 4 weeks, increase the dose to 5.0 mg once weekly.
- If after at least 4 weeks additional glycemic control is needed, increase dosage by 2.5mg until desired effects achieved.
- Maximum dosage = 15mg once weekly
- Administer with or without meals; any time of day.
For Weight Loss – SubQ
- Start at 2.5 mg SQ once weekly.
- After 4 weeks, increase the dose to 5.0 mg once weekly.
- Titrate until desired effects are seen; if stalls, then increase by 2.5mg weekly until maximum of 15mg once weekly is achieved.
- Administer with or without meals; any time of day.
Special Dosage Requirements:
- Renal Insufficiency: No dose adjustment is required in patients with renal impairment.
- Hepatic Insufficiency: No dose adjustment is recommended in patients with hepatic insufficiency
- Geriatrics (≥65 years): No dose adjustment is required in patients over 65 years of age
- Pediatrics (< 18 yrs): The safety and effectiveness of tirzepatide has not been studied in ages 18 and under. Tirzepatide is not recommended for pediatric use.
Safety and Potential Side Effects/Contraindications:
- Tirzepatide is reported safe in recommended dosages.
- Tirzepatide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
|
- Contraindicated in patients with hypersensitivity to Tirzepatide.
- Effectiveness of tirzepatide not impacted by age, gender, race, ethnicity, region, or by baseline BMI, HbA1c, diabetes duration, or renal function.
- Serious side effects can include:
- Pancreatitis: Has been reported in clinical trials. Discontinue promptly if pancreatitis is suspected.
- Hypoglycemia with Concomitant Use of Insulin Secretagogues or Insulin: Concomitant use with an insulin secretagogue or insulin may increase the risk of hypoglycemia, including severe hypoglycemia. Reducing dose of insulin secretagogue or insulin may be necessary.
- Hypersensitivity Reactions: Hypersensitivity reactions have been reported. Discontinue MOUNJARO if suspected.
- Acute Kidney Injury: Monitor renal function in patients with renal impairment reporting severe adverse gastrointestinal reactions
- Severe Gastrointestinal Disease: Use may be associated with gastrointestinal adverse reactions, sometimes severe. Has not been studied in patients with severe gastrointestinal disease and is not recommended in these patients.
- Diabetic Retinopathy Complications in Patients with a History of Diabetic Retinopathy: Has not been studied in patients with nonproliferative diabetic retinopathy requiring acute therapy, proliferative diabetic retinopathy, or diabetic macular edema. Monitor patients with a history of diabetic retinopathy for progression.
- Acute Gallbladder Disease: Has occurred in clinical trials. If cholelithiasis is suspected, gallbladder studies and clinical follow-up are indicated.
- The most common adverse reactions, reported in ≥5% of patients treated with tirzepatide are:
- Nausea/vomiting – nausea reported in 12-18%
- Diarrhea – reported in 12-17%
- Abdominal pain
- Belching
- Constipation
- Tirzepatide should not be used during pregnancy or breastfeeding.
- Tirzepatide delays gastric emptying – there is a potential to impact the absorption of concomitantly administered oral medications.
- Use of tirzepatide may reduce the efficacy of oral hormonal contraceptives. Advise patients using oral hormonal contraceptives to switch to a non-oral contraceptive method or add a barrier method of contraception for 4 weeks after initiation and for 4 weeks after each dose escalation with tirzepatide.
Description
The FDA approved tirzepatide as Munjaro® in May 2022 to improve glycemic control in adults with type 2 diabetes mellitus along with diet and exercise, and with off-label uses in obesity.
Tirzepatide is a first of class, long-acting once-weekly GIP and GLP-1 receptor agonist. It is a 39 amino acid peptide. The peptide component is engineered from the GIP sequence containing 2 non-coded amino acids (aminoisobutyric acid, Aib) in positions 2 and 13, a C-terminal amide, and Lys residue at position 20 is attached to 1,20-eicosanedioic acid via a linker. Tirzepatide enhances first- and second-phase insulin secretion, and reduces glucagon levels, both in a glucose-dependent manner.
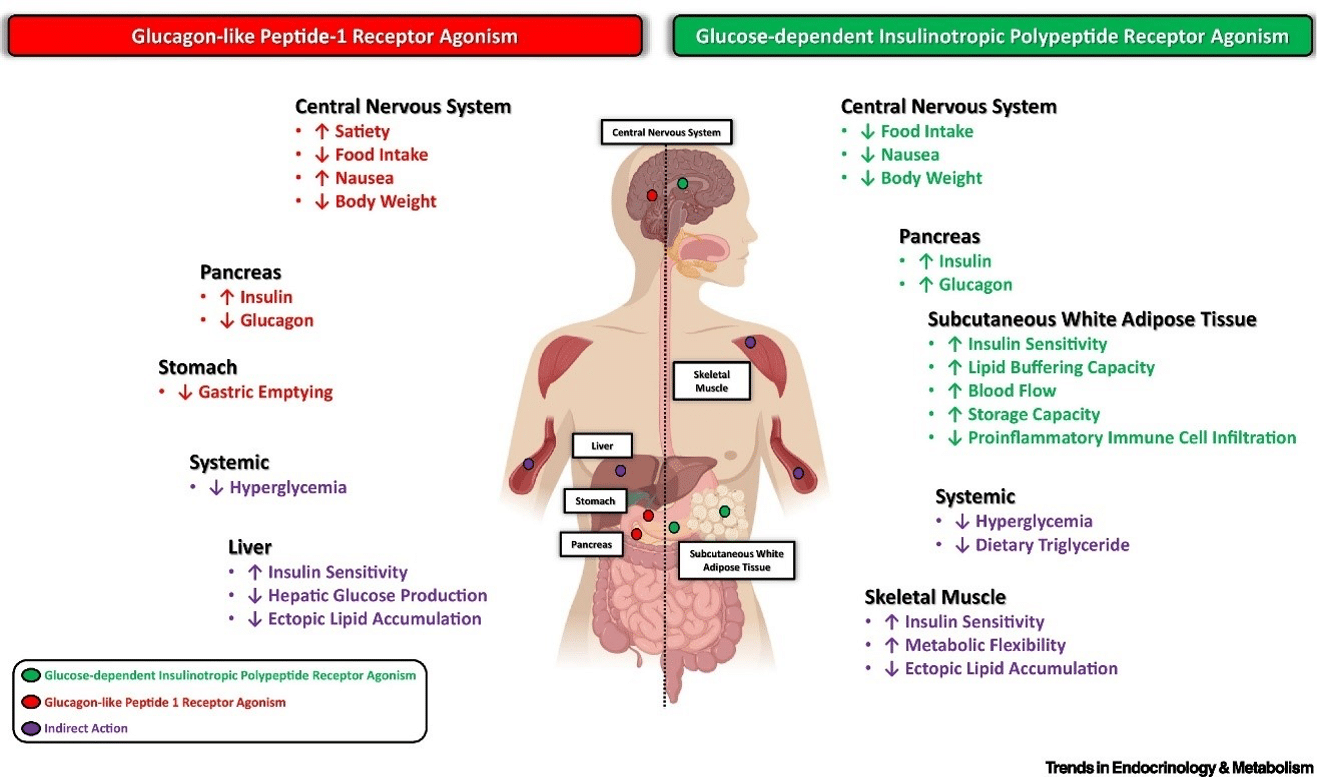
GIP and GLP1 are incretin hormones: they are released in the intestine in response to nutrient intake and stimulate pancreatic beta cell activity secreting insulin. GIP and GLP1 also have other metabolic functions. GLP1, in particular, reduces food intake and delays gastric emptying. Moreover, Tirzepatide has been shown to improve blood pressure and to reduce Low-Density Lipoprotein (LDL) cholesterol and triglycerides. Tirzepatide efficacy and safety were assessed in a phase III SURPASS 1–5 clinical trial program. Recently, the Food and Drug Administration approved Tirzepatide subcutaneous injections as monotherapy or combination therapy, with diet and physical exercise, to achieve better glycemic blood levels in patients with diabetes. Other clinical trials are currently underway to evaluate tirzepatide’s use in other diseases.
Tirzepatide has a greater affinity to GIP receptors than to GLP-1, and this dual agonist behavior has been shown to produce greater reductions of hyperglycemia compared to a selective GLP-1 receptor agonist.[i] Signaling studies reported that tirzepatide mimics the actions of natural GIP at the GIP receptor.[ii] At the GLP-1 receptor, though, tirzepatide shows bias towards cAMP (a messenger associated with regulation of glycogen, sugar, and lipid metabolism) generation, rather than beta-arrestin recruitment. This combination of preference towards GIP receptor and distinct signaling properties at GLP-1 suggest this biased agonism increases insulin secretion. [iii] Tirzepatide has been reported to increase levels of adiponectin, an adipokine involved in the regulation of both glucose and lipid metabolism with a maximum increase of 26% from baseline after 26 weeks, at the 10 mg dosage.[iv]
Tirzepatide’s mechanisms include:[v]
Tirzepatide research results report:
- Tirzepatide achieved up to 15.7% weight loss in adults with obesity or overweight and type 2 diabetes in SURMOUNT-2
- Average body weight reductions: 12.8% (10 mg), 14.7% (15 mg), 3.2% (placebo)
- Percentage of participants achieving body weight reductions of ≥5%: 79.2% (10 mg), 82.7% (15 mg), 32.5% (placebo)
- Percentage of participants achieving body weight reductions of ≥15%: 39.7% (10 mg), 48.0% (15 mg), 2.7% (placebo)
- Tirzepatide reported to decrease HbA1c by an average of 2.4% after 6 months therapy.
Research Summary in Treatment of Obesity*
- Jastreboff AM, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205-16.
Tirzepatide is also being studied as a potential treatment for people with obesity and/or overweight with heart failure with preserved ejection fraction (HFpEF), obstructive sleep apnea (OSA), and non-alcoholic steatohepatitis (NASH). Studies of tirzepatide in chronic kidney disease (CKD) and in morbidity/mortality in obesity (MMO) are also ongoing.
Clinical Research
IPS Level of Evidence
IPS Clinical Pharmacists have developed a method of ranking the studies so that the practitioner can easily discern the level of evidence this study provides to the topic. Levels 1-8 are listed below:
| Level of Evidence | Description | |
| X | Level 1 | FDA Approved Drug studies |
| X | Level 2 | Evidence obtained from systematic review and/or meta-analyses of studies including RCTs and other human studies |
| X | Level 3 | Evidence obtained from a RCT |
| Level 4 | Evidence obtained from a study without randomization | |
| Level 5 | Evidence obtained from case reports | |
| X | Level 6 | Evidence obtained from in vitro human studies |
| Level 7 | Evidence obtained from laboratory animal studies | |
| X | Level 8 | Evidence obtained from Opinions or Reviews |
Levels
Level 1
Wilson JM, et al. The dual glucose‐dependent insulinotropic polypeptide and glucagon‐like peptide‐1 receptor agonist tirzepatide improves cardiovascular risk biomarkers in patients with type 2 diabetes: A post hoc analysis. Dibetes Obes Metab. 2022;24(1):148-53.
Abstract
Abstract: In a phase 2 trial of once‐weekly tirzepatide (1, 5, 10, or 15 mg), dulaglutide (1.5 mg), or placebo, the dual glucose‐dependent insulinotropic polypeptide and glucagon‐like peptide‐1 receptor agonist tirzepatide dose‐dependently reduced HbA1c and body weight in patients with type 2 diabetes. In this post hoc analysis, inflammation, endothelial dysfunction, and cellular stress biomarkers were measured at baseline, 4, 12, and 26 weeks to evaluate the additional effects of tirzepatide on cardiovascular risk factors. At 26 weeks, tirzepatide 10 and 15 mg decreased YKL‐40 (also known as chitinase‐3 like‐protein‐1), intercellular adhesion molecule 1 (ICAM‐1), leptin, and growth differentiation factor 15 levels versus baseline, and YKL‐40 and leptin levels versus placebo and dulaglutide. Tirzepatide 15 mg also decreased ICAM‐1 levels versus placebo and dulaglutide, and high‐sensitivity C‐reactive protein (hsCRP) levels versus baseline and placebo, but not dulaglutide. GlycA, interleukin 6, vascular cell adhesion molecule 1, and N‐terminal‐pro hormone B‐type natriuretic peptide levels were not significantly changed in any group. YKL‐40, hsCRP, and ICAM‐1 levels rapidly decreased within 4 weeks of treatment with tirzepatide 10 and 15 mg, whereas the decrease in leptin levels was more gradual and did not plateau by 26 weeks. In this hypothesis‐generating exploratory analysis, tirzepatide decreased several biomarkers that have been associated with cardiovascular risk.
![]() https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9292792/pdf/DOM-24-148.pdf
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9292792/pdf/DOM-24-148.pdf
Thomas MK, et al. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes.
Abstract
Abstract: Context: Novel dual glucose-dependent insulinotropic polypeptide (GIP) and glucagonlike peptide-1 (GLP-1) receptor agonist (RA) tirzepatide demonstrated substantially greater glucose control and weight loss (WL) compared with selective GLP-1RA dulaglutide. Objective: Explore mechanisms of glucose control by tirzepatide. Design: Post hoc analyses of fasting biomarkers and multiple linear regression analysis. Setting: Forty-seven sites in 4 countries. Patients or other Participants: Three hundred and sixteen subjects with type 2 diabetes. Interventions: Tirzepatide (1, 5, 10, 15 mg), dulaglutide (1.5 mg), placebo. Main Outcome Measures: Analyze biomarkers of beta-cell function and insulin resistance (IR) and evaluate WL contributions to IR improvements at 26 weeks. Results: Homeostatic model assessment (HOMA) 2-B significantly increased with dulaglutide and tirzepatide 5, 10, and 15 mg compared with placebo (P ≤ .02). Proinsulin/ insulin and proinsulin/C-peptide ratios significantly decreased with tirzepatide 10 and 15 mg compared with placebo and dulaglutide (P ≤ .007). Tirzepatide 10 and 15 mg significantly decreased fasting insulin (P ≤ .033) and tirzepatide 10 mg significantly decreased HOMA2-IR (P = .004) compared with placebo and dulaglutide. Markers of improved insulin sensitivity (IS) adiponectin, IGFBP-1, and IGFBP-2 significantly increased by 1 or more doses of Tirzepatide (P < 0.05) . To determine whether improvements in IR were directly attributable to WL, multiple linear regression analysis with potential confounding variables age, sex, metformin, triglycerides, and glycated hemoglobin A1c was conducted. WL significantly (P ≤ .028) explained only 13% and 21% of improvement in HOMA2-IR with tirzepatide 10 and 15 mg, respectively.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7823251/
Jastreboff AM, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205-16.
Abstract
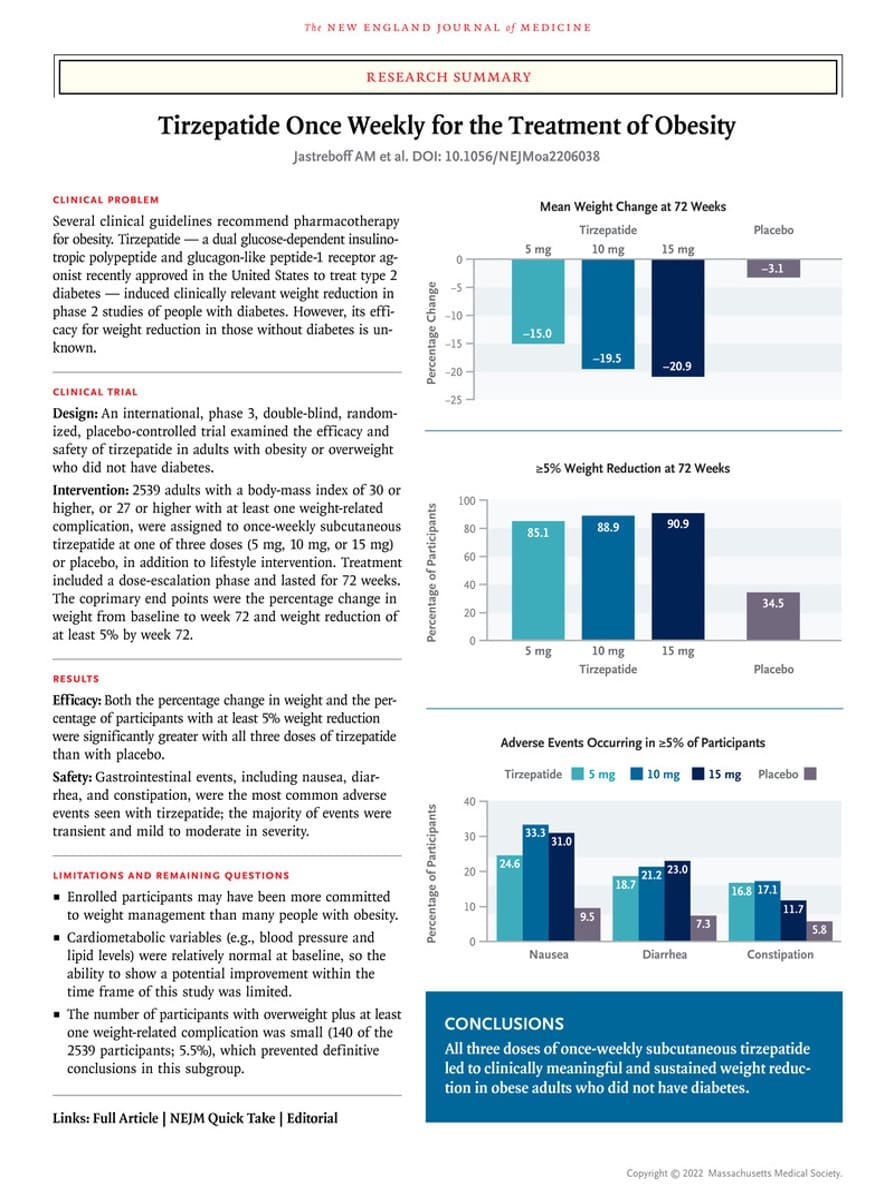
BACKGROUND: Obesity is a chronic disease that results in substantial global morbidity and mortality. The efficacy and safety of tirzepatide, a novel glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 receptor agonist, in people with obesity are not known.
METHODS: In this phase 3 double-blind, randomized, controlled trial, we assigned 2539 adults with a body-mass index (BMI; the weight in kilograms divided by the square of the height in meters) of 30 or more, or 27 or more and at least one weight-related complication, excluding diabetes, in a 1:1:1:1 ratio to receive once-weekly, subcutaneous tirzepatide (5 mg, 10 mg, or 15 mg) or placebo for 72 weeks, including a 20-week dose-escalation period. Coprimary end points were the percentage change in weight from baseline and a weight reduction of 5% or more. The treatment-regimen estimand assessed effects regardless of treatment discontinuation in the intention-to-treat population.
RESULTS: At baseline, the mean body weight was 104.8 kg, the mean BMI was 38.0, and 94.5% of participants had a BMI of 30 or higher. The mean percentage change in weight at week 72 was −15.0% (95% confidence interval [CI], −15.9 to −14.2) with 5-mg weekly doses of tirzepatide, −19.5% (95% CI, −20.4 to −18.5) with 10-mg doses, and −20.9% (95% CI, −21.8 to −19.9) with 15-mg doses and −3.1% (95% CI, −4.3 to −1.9) with placebo (P<0.001 for all comparisons with placebo). The percentage of participants who had weight reduction of 5% or more was 85% (95% CI, 82 to 89), 89% (95% CI, 86 to 92), and 91% (95% CI, 88 to 94) with 5 mg, 10 mg, and 15 mg of tirzepatide, respectively, and 35% (95% CI, 30 to 39) with placebo; 50% (95% CI, 46 to 54) and 57% (95% CI, 53 to 61) of participants in the 10-mg and 15-mg groups had a reduction in body weight of 20% or more, as compared with 3% (95% CI, 1 to 5) in the placebo group (P<0.001 for all comparisons with placebo). Improvements in all prespecified cardiometabolic measures were observed with tirzepatide. The most common adverse events with tirzepatide were gastrointestinal, and most were mild to moderate in severity, occurring primarily during dose escalation. Adverse events caused treatment discontinuation in 4.3%, 7.1%, 6.2%, and 2.6% of participants receiving 5-mg, 10-mg, and 15-mg tirzepatide doses and placebo, respectively.
CONCLUSIONS: In this 72-week trial in participants with obesity, 5 mg, 10 mg, or 15 mg of tirzepatide once weekly provided substantial and sustained reductions in body weight.
Rosenstock J, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-55.
Abstract
Background: Despite advancements in care, many people with type 2 diabetes do not meet treatment goals; thus, development of new therapies is needed. We aimed to assess efficacy, safety, and tolerability of novel dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist tirzepatide monotherapy versus placebo in people with type 2 diabetes inadequately controlled by diet and exercise alone.
Methods: We did a 40-week, double-blind, randomised, placebo-controlled, phase 3 trial (SURPASS-1), at 52 medical research centres and hospitals in India, Japan, Mexico, and the USA. Adult participants (≥18 years) were included if they had type 2 diabetes inadequately controlled by diet and exercise alone and if they were naive to injectable diabetes therapy. Participants were randomly assigned (1:1:1:1) via computer-generated random sequence to once a week tirzepatide (5, 10, or 15 mg), or placebo. All participants, investigators, and the sponsor were masked to treatment assignment. The primary endpoint was the mean change in glycated haemoglobin (HbA1c) from baseline at 40 weeks. This study is registered with ClinicalTrials.gov, NCT03954834.
Findings: From June 3, 2019, to Oct 28, 2020, of 705 individuals assessed for eligibility, 478 (mean baseline HbA1c 7·9% [63 mmol/mol], age 54·1 years [SD 11·9], 231 [48%] women, diabetes duration 4·7 years, and body-mass index 31·9 kg/m2) were randomly assigned to tirzepatide 5 mg (n=121 [25%]), tirzepatide 10 mg (n=121 [25%]), tirzepatide 15 mg (n=121 [25%]), or placebo (n=115 [24%]). 66 (14%) participants discontinued the study drug and 50 (10%) discontinued the study prematurely. At 40 weeks, all tirzepatide doses were superior to placebo for changes from baseline in HbA1c, fasting serum glucose, bodyweight, and HbA1c targets of less than 7·0% (<53 mmol/mol) and less than 5·7% (<39 mmol/mol). Mean HbA1c decreased from baseline by 1·87% (20 mmol/mol) with tirzepatide 5 mg, 1·89% (21 mmol/mol) with tirzepatide 10 mg, and 2·07% (23 mmol/mol) with tirzepatide 15 mg versus +0·04% with placebo (+0·4 mmol/mol), resulting in estimated treatment differences versus placebo of -1·91% (-21 mmol/mol) with tirzepatide 5 mg, -1·93% (-21 mmol/mol) with tirzepatide 10 mg, and -2·11% (-23 mmol/mol) with tirzepatide 15 mg (all p<0·0001). More participants on tirzepatide than on placebo met HbA1c targets of less than 7·0% (<53 mmol/mol; 87-92% vs 20%) and 6·5% or less (≤48 mmol/mol; 81-86% vs 10%) and 31-52% of patients on tirzepatide versus 1% on placebo reached an HbA1c of less than 5·7% (<39 mmol/mol). Tirzepatide induced a dose-dependent bodyweight loss ranging from 7·0 to 9·5 kg. The most frequent adverse events with tirzepatide were mild to moderate and transient gastrointestinal events, including nausea (12-18% vs 6%), diarrhoea (12-14% vs 8%), and vomiting (2-6% vs 2%). No clinically significant (<54 mg/dL [<3 mmol/L]) or severe hypoglycaemia were reported with tirzepatide. One death occurred in the placebo group.
Interpretation: Tirzepatide showed robust improvements in glycaemic control and bodyweight, without increased risk of hypoglycaemia. The safety profile was consistent with GLP-1 receptor agonists, indicating a potential monotherapy use of tirzepatide for type 2 diabetes treatment.
https://www.sciencedirect.com/science/article/abs/pii/S0140673621013246
Frias JP, et al. Tirzepatide versus Semaglutide once weekly in patients with Type 2 diabetes. NEJM. 2021;385(6):503-15.
Abstract
Background: Tirzepatide is a dual glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 (GLP-1) receptor agonist that is under development for the treatment of type 2 diabetes. The efficacy and safety of once-weekly tirzepatide as compared with semaglutide, a selective GLP-1 receptor agonist, are unknown.
Methods: In an open-label, 40-week, phase 3 trial, we randomly assigned 1879 patients, in a 1:1:1:1 ratio, to receive tirzepatide at a dose of 5 mg, 10 mg, or 15 mg or semaglutide at a dose of 1 mg. At baseline, the mean glycated hemoglobin level was 8.28%, the mean age 56.6 years, and the mean weight 93.7 kg. The primary end point was the change in the glycated hemoglobin level from baseline to 40 weeks.
Results: The estimated mean change from baseline in the glycated hemoglobin level was -2.01 percentage points, -2.24 percentage points, and -2.30 percentage points with 5 mg, 10 mg, and 15 mg of tirzepatide, respectively, and -1.86 percentage points with semaglutide; the estimated differences between the 5-mg, 10-mg, and 15-mg tirzepatide groups and the semaglutide group were -0.15 percentage points (95% confidence interval [CI], -0.28 to -0.03; P = 0.02), -0.39 percentage points (95% CI, -0.51 to -0.26; P<0.001), and -0.45 percentage points (95% CI, -0.57 to -0.32; P<0.001), respectively. Tirzepatide at all doses was noninferior and superior to semaglutide. Reductions in body weight were greater with tirzepatide than with semaglutide (least-squares mean estimated treatment difference, -1.9 kg, -3.6 kg, and -5.5 kg, respectively; P<0.001 for all comparisons). The most common adverse events were gastrointestinal and were primarily mild to moderate in severity in the tirzepatide and semaglutide groups (nausea, 17 to 22% and 18%; diarrhea, 13 to 16% and 12%; and vomiting, 6 to 10% and 8%, respectively). Of the patients who received tirzepatide, hypoglycemia (blood glucose level, <54 mg per deciliter) was reported in 0.6% (5-mg group), 0.2% (10-mg group), and 1.7% (15-mg group); hypoglycemia was reported in 0.4% of those who received semaglutide. Serious adverse events were reported in 5 to 7% of the patients who received tirzepatide and in 3% of those who received semaglutide.
Conclusions: In patients with type 2 diabetes, tirzepatide was noninferior and superior to semaglutide with respect to the mean change in the glycated hemoglobin level from baseline to 40 weeks.
Ludvik B, et al. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial. Lancet. 2021;398(10300):583-98.
Abstract
Background: Tirzepatide is a novel dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist under development for the treatment of type 2 diabetes. We aimed to assess the efficacy and safety of tirzepatide versus titrated insulin degludec in people with type 2 diabetes inadequately controlled by metformin with or without SGLT2 inhibitors.
Methods: In this open-label, parallel-group, multicentre (122 sites), multinational (13 countries), phase 3 study, eligible participants (aged ≥18 years) had a baseline glycated haemoglobin (HbA1c) of 7·0-10·5%, body-mass index of at least 25 kg/m2, stable weight, and were insulin-naive and treated with metformin alone or in combination with an SGLT2 inhibitor for at least 3 months before screening. Participants were randomly assigned (1:1:1:1), using an interactive web-response system, to once-weekly subcutaneous injection of tirzepatide (5, 10, or 15 mg) or once-daily subcutaneous injection of titrated insulin degludec, and were stratified by country, HbA1c, and concomitant use of oral antihyperglycaemic medications. Tirzepatide was initially given at 2·5 mg and the dose was escalated by 2·5 mg every 4 weeks until the assigned dose was reached. Insulin degludec was initially given at 10 U per day and was titrated once weekly to a fasting self-monitored blood glucose of less than 5·0 mmol/L (<90 mg/dL), following a treat-to-target algorithm, for 52 weeks. The primary efficacy endpoint was non-inferiority of tirzepatide 10 mg or 15 mg, or both, versus insulin degludec in mean change from baseline in HbA1c at week 52. Key secondary efficacy endpoints were non-inferiority of tirzepatide 5 mg versus insulin degludec in mean change from baseline in HbA1c at week 52, superiority of all doses of tirzepatide versus insulin degludec in mean change from baseline in HbA1c and bodyweight, and the proportion of participants achieving HbA1c of less than 7·0% (<53 mmol/mol) at week 52. We used a boundary of 0·3% to establish non-inferiority in HbA1c difference between treatments. Efficacy and safety analyses were assessed in the modified intention-to-treat population (all participants who received at least one dose of study drug). This trial is registered with ClinicalTrials.gov, number NCT03882970, and is complete.
Findings: Between April 1 and Nov 15, 2019, we assessed 1947 participants for eligibility, 1444 of whom were randomly assigned to treatment. The modified intention-to-treat population was 1437 participants from the tirzepatide 5 mg (n=358), tirzepatide 10 mg (n=360), tirzepatide 15 mg (n=359), and insulin degludec (n=360) groups. From a mean baseline HbA1c of 8·17% (SD 0·91), the reductions in HbA1c at week 52 were 1·93% (SE 0·05) for tirzepatide 5 mg, 2·20% (0·05) for tirzepatide 10 mg, and 2·37% (0·05) for tirzepatide 15 mg, and 1·34% (0·05) for insulin degludec. The non-inferiority margin of 0·3% was met. The estimated treatment difference (ETD) versus insulin degludec ranged from -0·59% to -1·04% for tirzepatide (p<0·0001 for all tirzepatide doses). The proportion of participants achieving a HbA1c of less than 7·0% (<53 mmol/mol) at week 52 was greater (p<0·0001) in all three tirzepatide groups (82%-93%) versus insulin degludec (61%). At week 52, from a baseline of 94·3 kg (SD 20·1), all three tirzepatide doses decreased bodyweight (-7·5 kg to -12·9 kg), whereas insulin degludec increased bodyweight by 2·3 kg. The ETD versus insulin degludec ranged from -9·8 kg to -15·2 kg for tirzepatide (p<0·0001 for all tirzepatide doses). The most common adverse events in tirzepatide-treated participants were mild to moderate gastrointestinal events that decreased over time. A higher incidence of nausea (12-24%), diarrhoea (15-17%), decreased appetite (6-12%), and vomiting (6-10%) was reported in participants treated with tirzepatide than in those treated with insulin degludec (2%, 4%, 1%, and 1%, respectively). Hypoglycaemia (<54 mg/dL or severe) was reported in five (1%), four (1%), and eight (2%) participants on tirzepatide 5, 10, and 15 mg, respectively, versus 26 (7%) on insulin degludec. Treatment discontinuation due to an adverse event was more common in the tirzepatide groups than in the insulin degludec group. Five participants died during the study; none of the deaths were considered by the investigators to be related to the study treatment.
Interpretation: In patients with type 2 diabetes, tirzepatide (5, 10, and 15 mg) was superior to titrated insulin degludec, with greater reductions in HbA1c and bodyweight at week 52 and a lower risk of hypoglycaemia. Tirzepatide showed a similar safety profile to that of GLP-1 receptor agonists.
https://endocrinologia.org.mx/pdf_socios/115_Once_weekly_tirzepatide_versus_Lancet_2021.pdf
Dahl D, et al. Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial. JAMA. 2022;327(6):534-45.
Abstract
Importance: The effects of tirzepatide, a dual glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 receptor agonist, as an addition to insulin glargine for treatment of type 2 diabetes have not been described.
Objective: To assess the efficacy and safety of tirzepatide added to insulin glargine in patients with type 2 diabetes with inadequate glycemic control.
Design, setting, and participants: Randomized phase 3 clinical trial conducted at 45 medical research centers and hospitals in 8 countries (enrollment from August 30, 2019, to March 20, 2020; follow-up completed January 13, 2021) in 475 adults with type 2 diabetes and inadequate glycemic control while treated with once-daily insulin glargine with or without metformin.
Interventions: Patients were randomized in a 1:1:1:1 ratio to receive once-weekly subcutaneous injections of 5-mg (n = 116), 10-mg (n = 119), or 15-mg (n = 120) tirzepatide or volume-matched placebo (n = 120) over 40 weeks. Tirzepatide was initiated at 2.5 mg/week and escalated by 2.5 mg every 4 weeks until the assigned dose was achieved.
Main outcomes and measures: The primary end point was mean change from baseline in glycated hemoglobin A1c (HbA1c) at week 40. The 5 key secondary end points included mean change in body weight and percentage of patients achieving prespecified HbA1c levels.
Results: Among 475 randomized participants (211 [44%] women; mean [SD] age, 60.6 [9.9] years; mean [SD] HbA1c, 8.31% [0.85%]), 451 (94.9%) completed the trial. Treatment was prematurely discontinued by 10% of participants in the 5-mg tirzepatide group, 12% in the 10-mg tirzepatide group, 18% in the 15-mg tirzepatide group, and 3% in the placebo group. At week 40, mean HbA1c change from baseline was -2.40% with 10-mg tirzepatide and -2.34% with 15-mg tirzepatide vs -0.86% with placebo (10 mg: difference vs placebo, -1.53% [97.5% CI, -1.80% to -1.27%]; 15 mg: difference vs placebo, -1.47% [97.5% CI, -1.75% to -1.20%]; P < .001 for both). Mean HbA1c change from baseline was -2.11% with 5-mg tirzepatide (difference vs placebo, -1.24% [95% CI, -1.48% to -1.01%]; P < .001]). Mean body weight change from baseline was -5.4 kg with 5-mg tirzepatide, -7.5 kg with 10-mg tirzepatide, -8.8 kg with 15-mg tirzepatide and 1.6 kg with placebo (5 mg: difference, -7.1 kg [95% CI, -8.7 to -5.4]; 10 mg: difference, -9.1 kg [95% CI, -10.7 to -7.5]; 15 mg: difference, -10.5 kg [95% CI, -12.1 to -8.8]; P < .001 for all). Higher percentages of patients treated with tirzepatide vs those treated with placebo had HbA1c less than 7% (85%-90% vs 34%; P < .001 for all). The most common treatment-emergent adverse events in the tirzepatide groups vs placebo group were diarrhea (12%-21% vs 10%) and nausea (13%-18% vs 3%).
Conclusions and relevance: Among patients with type 2 diabetes and inadequate glycemic control despite treatment with insulin glargine, the addition of subcutaneous tirzepatide, compared with placebo, to titrated insulin glargine resulted in statistically significant improvements in glycemic control after 40 weeks.
Level 2
Min T, Bain SC. The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Ther. 2021;12(1):143-57.
Abstract
Glucagon-like peptide 1 (GLP-1) based therapy is an established treatment option for the management of type 2 diabetes mellitus (T2DM) and is recommended early in the treatment algorithm owing to glycaemic efficacy, weight reduction and favourable cardiovascular outcomes. Glucose-dependent insulinotropic polypeptide (GIP), on the other hand, was thought to have no potential as a glucose-lowering therapy because of observations showing no insulinotropic effect from supraphysiological infusion in people with T2DM. However, emerging evidence has illustrated that co-infusion of GLP-1 and GIP has a synergetic effect, resulting in significantly increased insulin response and glucagonostatic response, compared with separate administration of each hormone. These observations have led to the development of a dual GIP/GLP-1 receptor agonist, known as a ‘twincretin’. Tirzepatide is a novel dual GIP/GLP-1 receptor agonist formulated as a synthetic peptide containing 39 amino acids, based on the native GIP sequence. Pre-clinical trials and phase 1 and 2 clinical trials indicate that tirzepatide has potent glucose lowering and weight loss with adverse effects comparable to those of established GLP-1 receptor agonists. The long-term efficacy, safety and cardiovascular outcomes of tirzepatide will be investigated in the SURPASS phase 3 clinical trial programme. In this paper, we will review the pre-clinical and phase 1 and 2 trials for tirzepatide in the management of T2DM and give an overview of the SURPASS clinical trials.
![]() https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7843845/pdf/13300_2020_Article_981.pdf
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7843845/pdf/13300_2020_Article_981.pdf
Forzano I, et al. Tirzepatide; a systematic update: Int J Mol Sci. 2022;23(23):14631.
Abstract
Tirzepatide is a new molecule capable of controlling glucose blood levels by combining the dual agonism of Glucose-Dependent Insulinotropic Polypeptide (GIP) and Glucagon-Like Peptide-1 (GLP-1) receptors. GIP and GLP1 are incretin hormones: they are released in the intestine in response to nutrient intake and stimulate pancreatic beta cell activity secreting insulin. GIP and GLP1 also have other metabolic functions. GLP1, in particular, reduces food intake and delays gastric emptying. Moreover, Tirzepatide has been shown to improve blood pressure and to reduce Low-Density Lipoprotein (LDL) cholesterol and triglycerides. Tirzepatide efficacy and safety were assessed in a phase III SURPASS 1–5 clinical trial program. Recently, the Food and Drug Administration approved Tirzepatide subcutaneous injections as monotherapy or combination therapy, with diet and physical exercise, to achieve better glycemic blood levels in patients with diabetes. Other clinical trials are currently underway to evaluate its use in other diseases. The scientific interest toward this novel, first-in-class medication is rapidly increasing. In this comprehensive and systematic review, we summarize the main results of the clinical trials investigating Tirzepatide and the currently available meta-analyses, emphasizing novel insights into its adoption in clinical practice for diabetes and its future potential applications in cardiovascular medicine.
https://www.mdpi.com/1422-0067/23/23/14631
Lin F, et al. Weight loss efficacy and safety of Tirzepatide: a systematic review. PLoS One. 2023;18(5):e0285197.
Abstract
Objective: Tirzeptide is a novel glucagon-like peptide-1 receptor (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) drug, which shows good efficiency for weight loss. Therefore, we aim to investigate the efficacy and safety of tirzepatide for weight loss in type 2 diabetes mellitus (T2DM) and obesity patients in this meta-analysis study.
Methods: Cochrane Library, PubMed, Embase, Clinical Trials, and Web of Science were searched from inception to October 5, 2022. All randomized controlled trials (RCTs) were included. The odds ratio (OR) was calculated using fixed-effects or random-effects models by Review Manager 5.3 software.
Results: In total, ten studies (12 reports) involving 9,873 patients were identified. A significant loss body weight in the tirzepatide group versus the placebo by -9.81 kg (95% CI (-12.09, -7.52), GLP-1 RAs by -1.05 kg (95% CI (-1.48, -0.63), and insulin by -1.93 kg (95% CI (-2.81, -1.05), respectively. In sub-analysis, the body weight of patients was significantly reduced in three tirzepatide doses (5 mg, 10 mg, and 15 mg) when compared with those of the placebo/GLP-1 RA/insulin. In terms of safety, the incidence of any adverse events and adverse events leading to study drug discontinuation was higher in the tirzepatide group, but the incidence of serious adverse events and hypoglycaemia was lower. Additionally, the gastrointestinal adverse events (including diarrhea, nausea, vomiting and decreased appetite) of tirzepatide were higher than those of placebo/basal insulin, but similar to GLP-1 RAs.
Conclusion: In conclusion, tirzeptide can significantly reduce the weight of T2DM and patient with obesity, and it is a potential therapeutic regimen for weight-loss, but we need to be vigilant about its gastrointestinal reaction.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10159347/
Karagiannis T, et al. Management of type 2 diabetes with the dual GIP/GLP-1 receptor agonist tirzepatide: a systematic review and meta-analysis. Diabetologia. 2022;65(8):1251-61.
Abstract
Aims/hypothesis: Tirzepatide is a novel dual glucose-dependent insulinotropic peptide (GIP) and glucagon-like peptide-1 receptor agonist (GLP-1 RA) currently under review for marketing approval. Individual trials have assessed the clinical profile of tirzepatide vs different comparators. We conducted a systematic review and meta-analysis to assess the efficacy and safety of tirzepatide for type 2 diabetes.
Methods: We searched PubMed, Embase, Cochrane and ClinicalTrials.gov up until 27 October 2021 for randomised controlled trials with a duration of at least 12 weeks that compared once-weekly tirzepatide 5, 10 or 15 mg with placebo or other glucose-lowering drugs in adults with type 2 diabetes irrespective of their background glucose-lowering treatment. The primary outcome was change in HbA1c from baseline. Secondary efficacy outcomes included change in body weight, proportion of individuals reaching the HbA1c target of <53 mmol/mol (<7.0%), ≤48 mmol/mol (≤6.5%) or <39 mmol/mol (<5.7%), and proportion of individuals with body weight loss of at least 5%, 10% or 15%. Safety outcomes included hypoglycaemia, gastrointestinal adverse events, treatment discontinuation due to adverse events, serious adverse events, and mortality. We used version 2 of the Cochrane risk-of-bias tool for randomised trials to assess risk of bias for the primary outcome.
Results: Seven trials (6609 participants) were included. A dose-dependent superiority in lowering HbA1c was evident with all three tirzepatide doses vs all comparators, with mean differences ranging from -17.71 mmol/mol (-1.62%) to -22.35 mmol/mol (-2.06%) vs placebo, -3.22 mmol/mol (-0.29%) to -10.06 mmol/mol (-0.92%) vs GLP-1 RAs, and -7.66 mmol/mol (-0.70%) to -12.02 mmol/mol (-1.09%) vs basal insulin regimens. Tirzepatide was more efficacious in reducing body weight; reductions vs GLP-1 RAs ranged from 1.68 kg with tirzepatide 5 mg to 7.16 kg with tirzepatide 15 mg. Incidence of hypoglycaemia with tirzepatide was similar vs placebo and lower vs basal insulin. Nausea was more frequent with tirzepatide vs placebo, especially with tirzepatide 15 mg (OR 5.60 [95% CI 3.12, 10.06]), associated with higher incidence of vomiting (OR 5.50 [95% CI 2.40, 12.59]) and diarrhoea (OR 3.31 [95% CI 1.40, 7.85]). Odds of gastrointestinal events were similar between tirzepatide and GLP-1 RAs, except for diarrhoea with tirzepatide 10 mg (OR 1.51 [95% CI 1.07, 2.15]). Tirzepatide 15 mg led to higher discontinuation rate of study medication due to adverse events regardless of comparator, while all tirzepatide doses were safe in terms of serious adverse events and mortality.
Conclusions/interpretation: A dose-dependent superiority on glycaemic efficacy and body weight reduction was evident with tirzepatide vs placebo, GLP-1 RAs and basal insulin. Tirzepatide did not increase the odds of hypoglycaemia but was associated with increased incidence of gastrointestinal adverse events. Study limitations include presence of statistical heterogeneity in the meta-analyses for change in HbA1c and body weight, assessment of risk of bias solely for the primary outcome, and generalisation of findings mainly to individuals who are overweight or obese and already on metformin-based background therapy.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9112245/pdf/125_2022_Article_5715.pdf
Level 3
Thomas MK, et al. Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes. J Clin Endocrinol Metab. 2021;106(2):388-96.
Abstract
Context: Novel dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist (RA) tirzepatide demonstrated substantially greater glucose control and weight loss (WL) compared with selective GLP-1RA dulaglutide.
Objective: Explore mechanisms of glucose control by tirzepatide.
Design: Post hoc analyses of fasting biomarkers and multiple linear regression analysis.
Setting: Forty-seven sites in 4 countries.
Patients or other participants: Three hundred and sixteen subjects with type 2 diabetes.
Interventions: Tirzepatide (1, 5, 10, 15 mg), dulaglutide (1.5 mg), placebo.
Main outcome measures: Analyze biomarkers of beta-cell function and insulin resistance (IR) and evaluate WL contributions to IR improvements at 26 weeks.
Results: Homeostatic model assessment (HOMA) 2-B significantly increased with dulaglutide and tirzepatide 5, 10, and 15 mg compared with placebo (P ≤ .02). Proinsulin/insulin and proinsulin/C-peptide ratios significantly decreased with tirzepatide 10 and 15 mg compared with placebo and dulaglutide (P ≤ .007). Tirzepatide 10 and 15 mg significantly decreased fasting insulin (P ≤ .033) and tirzepatide 10 mg significantly decreased HOMA2-IR (P = .004) compared with placebo and dulaglutide. Markers of improved insulin sensitivity (IS) adiponectin, IGFBP-1, and IGFBP-2 significantly increased by 1 or more doses of tirzepatide (P < .05). To determine whether improvements in IR were directly attributable to WL, multiple linear regression analysis with potential confounding variables age, sex, metformin, triglycerides, and glycated hemoglobin A1c was conducted. WL significantly (P ≤ .028) explained only 13% and 21% of improvement in HOMA2-IR with tirzepatide 10 and 15 mg, respectively.
Conclusions: Tirzepatide improved markers of IS and beta-cell function to a greater extent than dulaglutide. IS effects of tirzepatide were only partly attributable to WL, suggesting dual receptor agonism confers distinct mechanisms of glycemic control.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7823251/
Sattar N, et al. Tirzepatide cardiovascular event risk assessment: a pre-specified meta-analysis. Nat Med. 2022;28(3):591-98.
Abstract
Tirzepatide is a novel, once weekly, dual GIP/GLP-1 receptor agonist and is under development for the treatment of type 2 diabetes (T2D) and obesity. Its association with cardiovascular outcomes requires evaluation. This pre-specified cardiovascular meta-analysis included all seven randomized controlled trials with a duration of at least 26 weeks from the tirzepatide T2D clinical development program, SURPASS. The pre-specified primary objective of this meta-analysis was the comparison of the time to first occurrence of confirmed four-component major adverse cardiovascular events (MACE-4; cardiovascular death, myocardial infarction, stroke and hospitalized unstable angina) between pooled tirzepatide groups and control groups. A stratified Cox proportional hazards model, with treatment as a fixed effect and trial-level cardiovascular risk as the stratification factor, was used for the estimation of hazard ratios (HRs) and confidence intervals (CIs) comparing tirzepatide to control. Data from 4,887 participants treated with tirzepatide and 2,328 control participants were analyzed. Overall, 142 participants, 109 from the trial with high cardiovascular risk and 33 from the six trials with lower cardiovascular risk, had at least one MACE-4 event. The HRs comparing tirzepatide versus controls were 0.80 (95% CI, 0.57-1.11) for MACE-4; 0.90 (95% CI, 0.50-1.61) for cardiovascular death; and 0.80 (95% CI, 0.51-1.25) for all-cause death. No evidence of effect modifications was observed for any subgroups, although the evidence was stronger for participants with high cardiovascular risk. Tirzepatide did not increase the risk of major cardiovascular events in participants with T2D versus controls.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8938269/pdf/41591_2022_Article_1707.pdf
Coskun T, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14.
Abstract
Objective: A novel dual GIP and GLP-1 receptor agonist, LY3298176, was developed to determine whether the metabolic action of GIP adds to the established clinical benefits of selective GLP-1 receptor agonists in type 2 diabetes mellitus (T2DM).
Methods: LY3298176 is a fatty acid modified peptide with dual GIP and GLP-1 receptor agonist activity designed for once-weekly subcutaneous administration. LY3298176 was characterised in vitro, using signaling and functional assays in cell lines expressing recombinant or endogenous incretin receptors, and in vivo using body weight, food intake, insulin secretion and glycemic profiles in mice. A Phase 1, randomised, placebo-controlled, double-blind study was comprised of three parts: a single-ascending dose (SAD; doses 0.25-8 mg) and 4-week multiple-ascending dose (MAD; doses 0.5-10 mg) studies in healthy subjects (HS), followed by a 4-week multiple-dose Phase 1 b proof-of-concept (POC; doses 0.5-15 mg) in patients with T2DM (ClinicalTrials.gov no. NCT02759107). Doses higher than 5 mg were attained by titration, dulaglutide (DU) was used as a positive control. The primary objective was to investigate safety and tolerability of LY3298176.
Results: LY3298176 activated both GIP and GLP-1 receptor signaling in vitro and showed glucose-dependent insulin secretion and improved glucose tolerance by acting on both GIP and GLP-1 receptors in mice. With chronic administration to mice, LY3298176 potently decreased body weight and food intake; these effects were significantly greater than the effects of a GLP-1 receptor agonist. A total of 142 human subjects received at least 1 dose of LY3298176, dulaglutide, or placebo. The PK profile of LY3298176 was investigated over a wide dose range (0.25-15 mg) and supports once-weekly administration. In the Phase 1 b trial of diabetic subjects, LY3298176 doses of 10 mg and 15 mg significantly reduced fasting serum glucose compared to placebo (least square mean [LSM] difference [95% CI]: -49.12 mg/dL [-78.14, -20.12] and -43.15 mg/dL [-73.06, -13.21], respectively). Reductions in body weight were significantly greater with the LY3298176 1.5 mg, 4.5 mg and 10 mg doses versus placebo in MAD HS (LSM difference [95% CI]: -1.75 kg [-3.38, -0.12], -5.09 kg [-6.72, -3.46] and -4.61 kg [-6.21, -3.01], respectively) and doses of 10 mg and 15 mg had a relevant effect in T2DM patients (LSM difference [95% CI]: -2.62 kg [-3.79, -1.45] and -2.07 kg [-3.25, -0.88], respectively. The most frequent side effects reported with LY3298176 were gastrointestinal (vomiting, nausea, decreased appetite, diarrhoea, and abdominal distension) in both HS and patients with T2DM; all were dose-dependent and considered mild to moderate in severity.
Conclusions: Based on these results, the pharmacology of LY3298176 translates from preclinical to clinical studies. LY3298176 has the potential to deliver clinically meaningful improvement in glycaemic control and body weight. The data warrant further clinical evaluation of LY3298176 for the treatment of T2DM and potentially obesity.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6308032/pdf/main.pdf
Hartman ML, et al. Effects of Novel Dual GIP and GLP-1 Receptor Agonist Tirzepatide on Biomarkers of Nonalcoholic Steatohepatitis in Patients With Type 2 Diabetes. Diabetes Care. 2020;43(6):1352-55.
Abstract
Objective: To determine the effect of tirzepatide, a dual agonist of glucose-dependent insulinotropic polypeptide and glucagon-like peptide 1 receptors, on biomarkers of nonalcoholic steatohepatitis (NASH) and fibrosis in patients with type 2 diabetes mellitus (T2DM).
Research design and methods: Patients with T2DM received either once weekly tirzepatide (1, 5, 10, or 15 mg), dulaglutide (1.5 mg), or placebo for 26 weeks. Changes from baseline in alanine aminotransferase (ALT), aspartate aminotransferase (AST), keratin-18 (K-18), procollagen III (Pro-C3), and adiponectin were analyzed in a modified intention-to-treat population.
Results: Significant (P < 0.05) reductions from baseline in ALT (all groups), AST (all groups except tirzepatide 10 mg), K-18 (tirzepatide 5, 10, 15 mg), and Pro-C3 (tirzepatide 15 mg) were observed at 26 weeks. Decreases with tirzepatide were significant compared with placebo for K-18 (10 mg) and Pro-C3 (15 mg) and with dulaglutide for ALT (10, 15 mg). Adiponectin significantly increased from baseline with tirzepatide compared with placebo (10, 15 mg).
Conclusions: In post hoc analyses, higher tirzepatide doses significantly decreased NASH-related biomarkers and increased adiponectin in patients with T2DM.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7245348/pdf/dc191892.pdf
Level 7
Samms RJ, et al. Tirzepatide induces thermogenic-like amino acid signature in brown adipose tissue. Mol Metab. 2022;64:101550.
Abstract
Objectives: Tirzepatide, a dual GIP and GLP-1 receptor agonist, delivered superior glycemic control and weight loss compared to selective GLP-1 receptor (GLP-1R) agonism in patients with type 2 diabetes (T2D). These results have fueled mechanistic studies focused on understanding how tirzepatide achieves its therapeutic efficacy. Recently, we found that treatment with tirzepatide improves insulin sensitivity in humans with T2D and obese mice in concert with a reduction in circulating levels of branched-chain amino (BCAAs) and keto (BCKAs) acids, metabolites associated with development of systemic insulin resistance (IR) and T2D. Importantly, these systemic effects were found to be coupled to increased expression of BCAA catabolic genes in thermogenic brown adipose tissue (BAT) in mice. These findings led us to hypothesize that tirzepatide may lower circulating BCAAs/BCKAs by promoting their catabolism in BAT.
Methods: To address this question, we utilized a murine model of diet-induced obesity and employed stable-isotope tracer studies in combination with metabolomic analyses in BAT and other tissues.
Results: Treatment with tirzepatide stimulated catabolism of BCAAs/BCKAs in BAT, as demonstrated by increased labeling of BCKA-derived metabolites, and increases in levels of byproducts of BCAA breakdown, including glutamate, alanine, and 3-hydroxyisobutyric acid (3-HIB). Further, chronic administration of tirzepatide increased levels of multiple amino acids in BAT that have previously been shown to be elevated in response to cold exposure. Finally, chronic treatment with tirzepatide led to a substantial increase in several TCA cycle intermediates (α-ketoglutarate, fumarate, and malate) in BAT.
Conclusions: These findings suggest that tirzepatide induces a thermogenic-like amino acid profile in BAT, an effect that may account for reduced systemic levels of BCAAs in obese IR mice.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9396640/pdf/main.pdf
Level 8
Chavda VP, et al. Tirzepatide, a new era of dual-targeted treatment for diabetes and obesity: a mini-review. Molecules. 2022;27(13):4315.
Abstract
The prevalence of obesity and diabetes is an increasing global problem, especially in developed countries, and is referred to as the twin epidemics. As such, advanced treatment approaches are needed. Tirzepatide, known as a ‘twincretin’, is a ‘first-in-class’ and the only dual glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP) receptor agonist, that can significantly reduce glycemic levels and improve insulin sensitivity, as well as reducing body weight by more than 20% and improving lipid metabolism. This novel anti-diabetic drug is a synthetic peptide analog of the human GIP hormone with a C20 fatty-diacid portion attached which, via acylation technology, can bind to albumin in order to provide a dose of the drug, by means of subcutaneous injection, once a week, which is appropriate to its a half-life of about five days. Tirzepatide, developed by Eli Lilly, was approved, under the brand name Mounjaro, by the United States Food and Drug Administration in May 2022. This started the ‘twincretin’ era of enormously important and appealing dual therapeutic options for diabetes and obesity, as well as advanced management of closely related cardiometabolic settings, which constitute the leading cause of morbidity, disability, and mortality worldwide. Herein, we present the key characteristics of tirzepatide in terms of synthesis, structure, and activity, bearing in mind its advantages and shortcomings. Furthermore, we briefly trace the evolution of this kind of medical agent and discuss the development of clinical studies.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9268041/pdf/molecules-27-04315.pdf
Del Prato S, et al.; SURPASS-4 Investigators . Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet. 2021;398(10313):1811-1824.
Abstract
Background: We aimed to assess efficacy and safety, with a special focus on cardiovascular safety, of the novel dual GIP and GLP-1 receptor agonist tirzepatide versus insulin glargine in adults with type 2 diabetes and high cardiovascular risk inadequately controlled on oral glucose-lowering medications.
Methods: This open-label, parallel-group, phase 3 study was done in 187 sites in 14 countries on five continents. Eligible participants, aged 18 years or older, had type 2 diabetes treated with any combination of metformin, sulfonylurea, or sodium-glucose co-transporter-2 inhibitor, a baseline glycated haemoglobin (HbA1c) of 7·5-10·5% (58-91 mmol/mol), body-mass index of 25 kg/m2 or greater, and established cardiovascular disease or a high risk of cardiovascular events. Participants were randomly assigned (1:1:1:3) via an interactive web-response system to subcutaneous injection of either once-per-week tirzepatide (5 mg, 10 mg, or 15 mg) or glargine (100 U/mL), titrated to reach fasting blood glucose of less than 100 mg/dL. The primary endpoint was non-inferiority (0·3% non-inferiority boundary) of tirzepatide 10 mg or 15 mg, or both, versus glargine in HbA1c change from baseline to 52 weeks. All participants were treated for at least 52 weeks, with treatment continued for a maximum of 104 weeks or until study completion to collect and adjudicate major adverse cardiovascular events (MACE). Safety measures were assessed over the full study period. This study was registered with ClinicalTrials.gov, NCT03730662.
Findings: Patients were recruited between Nov 20, 2018, and Dec 30, 2019. 3045 participants were screened, with 2002 participants randomly assigned to tirzepatide or glargine. 1995 received at least one dose of tirzepatide 5 mg (n=329, 17%), 10 mg (n=328, 16%), or 15 mg (n=338, 17%), or glargine (n=1000, 50%), and were included in the modified intention-to-treat population. At 52 weeks, mean HbA1c changes with tirzepatide were -2·43% (SD 0·05) with 10 mg and -2·58% (0·05) with 15 mg, versus -1·44% (0·03) with glargine. The estimated treatment difference versus glargine was -0·99% (multiplicity adjusted 97·5% CI -1·13 to -0·86) for tirzepatide 10 mg and -1·14% (-1·28 to -1·00) for 15 mg, and the non-inferiority margin of 0·3% was met for both doses. Nausea (12-23%), diarrhoea (13-22%), decreased appetite (9-11%), and vomiting (5-9%) were more frequent with tirzepatide than glargine (nausea 2%, diarrhoea 4%, decreased appetite <1%, and vomiting 2%, respectively); most cases were mild to moderate and occurred during the dose-escalation phase. The percentage of participants with hypoglycaemia (glucose <54 mg/dL or severe) was lower with tirzepatide (6-9%) versus glargine (19%), particularly in participants not on sulfonylureas (tirzepatide 1-3% vs glargine 16%). Adjudicated MACE-4 events (cardiovascular death, myocardial infarction, stroke, hospitalisation for unstable angina) occurred in 109 participants and were not increased on tirzepatide compared with glargine (hazard ratio 0·74, 95% CI 0·51-1·08). 60 deaths (n=25 [3%] tirzepatide; n=35 [4%] glargine) occurred during the study.
Interpretation: In people with type 2 diabetes and elevated cardiovascular risk, tirzepatide, compared with glargine, demonstrated greater and clinically meaningful HbA1c reduction with a lower incidence of hypoglycaemia at week 52. Tirzepatide treatment was not associated with excess cardiovascular risk.
No full text available
Samms RJ, et al. How May GIP Enhance the Therapeutic Efficacy of GLP-1? TGrends Endocrinol Metab. 2020;31(6):410-20.
Abstract
Glucagon-like peptide-1 (GLP-1) receptor agonists improve glucose homeostasis, reduce bodyweight, and over time benefit cardiovascular health in type 2 diabetes mellitus (T2DM). However, dose-related gastrointestinal effects limit efficacy, and therefore agents possessing GLP-1 pharmacology that can also target alternative pathways may expand the therapeutic index. One approach is to engineer GLP-1 activity into the sequence of glucose-dependent insulinotropic polypeptide (GIP). Although the therapeutic implications of the lipogenic actions of GIP are debated, its ability to improve lipid and glucose metabolism is especially evident when paired with the anorexigenic mechanism of GLP-1. We review the complexity of GIP in regulating adipose tissue function and energy balance in the context of recent findings in T2DM showing that dual GIP/GLP-1 receptor agonist therapy produces profound weight loss, glycemic control, and lipid lowering.
https://www.cell.com/action/showPdf?pii=S1043-2760%2820%2930048-5
Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
Abstract
Tirzepatide (LY3298176) is a dual GIP and GLP-1 receptor agonist under development for the treatment of type 2 diabetes mellitus (T2DM), obesity, and nonalcoholic steatohepatitis. Early phase trials in T2DM indicate that tirzepatide improves clinical outcomes beyond those achieved by a selective GLP-1 receptor agonist. Therefore, we hypothesized that the integrated potency and signaling properties of tirzepatide provide a unique pharmacological profile tailored for improving broad metabolic control. Here, we establish methodology for calculating occupancy of each receptor for clinically efficacious doses of the drug. This analysis reveals a greater degree of engagement of tirzepatide for the GIP receptor than the GLP-1 receptor, corroborating an imbalanced mechanism of action. Pharmacologically, signaling studies demonstrate that tirzepatide mimics the actions of native GIP at the GIP receptor but shows bias at the GLP-1 receptor to favor cAMP generation over β-arrestin recruitment, coincident with a weaker ability to drive GLP-1 receptor internalization compared with GLP-1. Experiments in primary islets reveal β-arrestin1 limits the insulin response to GLP-1, but not GIP or tirzepatide, suggesting that the biased agonism of tirzepatide enhances insulin secretion. Imbalance toward GIP receptor, combined with distinct signaling properties at the GLP-1 receptor, together may account for the promising efficacy of this investigational agent.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7526454/pdf/jciinsight-5-140532.pdf
Kim W, Egan M. The role of incretins in glucose homeostasis and diabetes treatment. Pharmacol Rev. 2008;60(4):470-12.
Abstract
Incretins are gut hormones that are secreted from enteroendocrine cells into the blood within minutes after eating. One of their many physiological roles is to regulate the amount of insulin that is secreted after eating. In this manner, as well as others to be described in this review, their final common raison d’être is to aid in disposal of the products of digestion. There are two incretins, known as glucose-dependent insulinotropic peptide (GIP) and glucagon-like peptide-1 (GLP-1), that share many common actions in the pancreas but have distinct actions outside of the pancreas. Both incretins are rapidly deactivated by an enzyme called dipeptidyl peptidase 4 (DPP4). A lack of secretion of incretins or an increase in their clearance are not pathogenic factors in diabetes. However, in type 2 diabetes (T2DM), GIP no longer modulates glucose-dependent insulin secretion, even at supraphysiological (pharmacological) plasma levels, and therefore GIP incompetence is detrimental to β-cell function, especially after eating. GLP-1, on the other hand, is still insulinotropic in T2DM, and this has led to the development of compounds that activate the GLP-1 receptor with a view to improving insulin secretion. Since 2005, two new classes of drugs based on incretin action have been approved for lowering blood glucose levels in T2DM: an incretin mimetic (exenatide, which is a potent long-acting agonist of the GLP-1 receptor) and an incretin enhancer (sitagliptin, which is a DPP4 inhibitor). Exenatide is injected subcutaneously twice daily and its use leads to lower blood glucose and higher insulin levels, especially in the fed state. There is glucose-dependency to its insulin secretory capacity, making it unlikely to cause low blood sugars (hypoglycemia). DPP4 inhibitors are orally active and they increase endogenous blood levels of active incretins, thus leading to prolonged incretin action. The elevated levels of GLP-1 are thought to be the mechanism underlying their blood glucose-lowering effects.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2696340/pdf/nihms86105.pdf
[1] Thomas MK, et al. “Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes”. The Journal of Clinical Endocrinology and Metabolism. 2021;106 (2):388–396.
[2] Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
[3] Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
[4] Thomas MK, et al. “Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes”. The Journal of Clinical Endocrinology and Metabolism. 2021;106 (2):388–396.
[5] Chavda VP, et al. Tirzepatide, a new era of dual-targeted treatment for diabetes and obesity: a mini-review. Molecules. 2022;27(13):4315.
[i] Thomas MK, et al. “Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes”. The Journal of Clinical Endocrinology and Metabolism. 2021;106 (2):388–396.
[ii] Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
[iii] Willard FS, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532.
[iv] Thomas MK, et al. “Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes”. The Journal of Clinical Endocrinology and Metabolism. 2021;106 (2):388–396.
[v] Chavda VP, et al. Tirzepatide, a new era of dual-targeted treatment for diabetes and obesity: a mini-review. Molecules. 2022;27(13):4315.